Adipic Acid-Based Polyester Diols

Ordinary aliphatic polyester polyols are actually mostly polyester diols, generally formed by the polycondensation of adipic acid (a small number of products use sebacic acid) with one or more diols such as ethylene glycol, propylene glycol, 1,4-butanediol, and diethylene glycol.
Polyesters prepared using mixed diols or mixed diacids have complex structural formulas. The following only shows the structural formula of polyester diols synthesized from adipic acid (AA) and ethylene glycol (or butanediol, etc.):
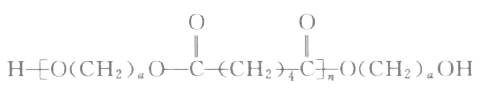
In the formula, O(CH2)αO represents the small molecule diol chain segment, where α = 2, 3, 4, 6, etc. —CO(CH2)4CO— represents the adipic acid chain segment.
Physicochemical Properties
Depending on the raw material composition, polyester diols are usually off-white waxy solids or colorless to light yellow viscous liquids at room temperature. Solid polyesters have a melting point of 25–50°C, and become viscous liquids after melting. They are slightly soluble in water. The acid value of polyesters is generally less than 1.0 mg KOH/g.
Polyester polyols are basically non-toxic. If they accidentally get into the eyes or on the skin, they should be immediately washed with tap water. Prolonged contact with the skin may cause mild irritation; it is best to wear safety glasses and gloves during operation.
The chemical industry standard HG/T 2707-1995 “Specifications for Polyester Polyols” specifies the physicochemical performance indicators of several adipic acid-based polyester polyols, using the polyester naming recommended by the industry standard. Currently, most manufacturers’ polyester color and acid value have met or exceeded these indicators.
In conventional naming, there is a corresponding English abbreviation, such as Polyethylene adipate glycol, abbreviated as PEA. The first P stands for “polymerization”; the last letter or the second to last letter A represents adipic acid (AA), I represents isophthalic acid, and P represents phthalic acid; the middle letters represent the diol component: E represents ethylene glycol (EG), D represents diethylene glycol (DEG), B represents 1,4-butanediol (BDO), P represents 1,2-propanediol (PG), H represents 1,6-hexanediol (HDO), N represents neopentyl glycol (NPG), M represents 2-methylpropanediol (MPD), T represents trimethylolpropane (TMP), and G represents glycerol (Gly). The number after the English abbreviation represents the molecular weight of the polyester.
Properties and Uses
Polyester-type polyurethanes have good polar groups such as ester and urethane groups, strong cohesive strength and adhesion, and high strength and wear resistance. Therefore, aliphatic polyester diols are mostly used in the production of castable polyurethane elastomers, thermoplastic polyurethane elastomers, microporous polyurethane shoe soles, PU leather resins, polyurethane adhesives, polyurethane inks and color pastes, and fabric coatings. Polyester diols obtained from adipic acid and 1,4-butanediol, 1,6-hexanediol, or ethylene glycol are waxy solids, and the resulting polyurethane elastomers have strong crystallinity and high initial tack; polyesters obtained from diols with side groups, such as PMA and PPA, are liquid at room temperature, soft, and used in inks, soft leather, etc. PMA has better hydrolysis resistance.
PREVIOUS:Aromatic Polyether Polyols
CATEGORIES
LATEST NEWS
- Diethanolamine
- Triethanolamine
- Methyl Glucoside
- Mannitol
- Sorbitol
- Xylitol
- Pentaerythritol
- Trihydroxyethyl isocyanurate
- Trimethylolethane
- Glycerin
CONTACT US
Tel:+86 18688691634
Email:sam@1688pu.com
Add:No. 91 Central East Rd, Luxi Village, Gaobu Town, Dongguan 523283, China.

