Aromatic Polyether Polyols

Aromatic polyether polyols generally refer to polyether polyols containing benzene rings. This does not include ethylene-grafted polymer polyols. Polyether polyols containing benzene rings have characteristics such as heat resistance and flame retardancy.
There are many ways to obtain aromatic polyether polyols. For example, in industry, aromatic amines such as toluenediamine and diphenylmethanediamine are used as initiators for the ring-opening polymerization of propylene oxide/ethylene oxide to obtain tetrahydroxy polyethers; initiators containing benzene rings are synthesized using phenol, formaldehyde, and secondary amines as raw materials, and then aromatic polyether polyols are synthesized; and bisphenol A (BPA) is used as an initiator to synthesize bisphenol A polyether polyols.
Aromatic Polyether Diols
Amine compounds containing two active hydrogens, such as aniline or substituted aniline, can be used as initiators to synthesize polyether diols. These tertiary amino polyether diol products are mainly used as chain extenders for elastomers. Other industrialized products include polyether diols with bisphenol A as an initiator.
The CAS numbers for bisphenol A/ethylene oxide polyether are 32492-61-8 and 29086-67-7. The English name is ethoxylated bisphenol A, etc., and the alternative name is bisphenol A polyoxyethylene ether.
The CAS numbers for bisphenol A/propylene oxide polyether (propoxylated bisphenol A, bisphenol A polyoxypropylene ether) are 37353-75-6 and 29694-85-7. The CAS numbers for bisphenol A/PO/EO copolymer (ethoxylated/propoxylated bisphenol A, bisphenol A polyoxyethylene-oxypropylene ether) are 62611-29-4, 52367-02-9, and 65324-64-3. Structural formula of Bisphenol A polyoxyethylene-ethylene ether:
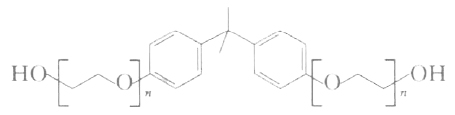
Hangzhou Bailang Additives Co., Ltd.’s toughening resin A-206 is bisphenol A polyoxyethylene ether (BPA+6EO), a colorless to light yellow transparent liquid with a hydroxyl value of 220-230 mg KOH/g, an acid value of ≤0.1 mg KOH/g, and potassium and sodium ions ≤15 mg/kg. Guodu Chemical (Kunshan) Co., Ltd.’s BP-11 is a polyether initiated with bisphenol A, with a hydroxyl value of 280-300 mg KOH/g, a viscosity of 800-2200 (60℃), a color of ≤100, and an acid value of ≤0.2 mg KOH/g. Jiangsu Hai’an Petrochemical Plant and Shenyang Purixing Fine Chemical Co., Ltd. also produce bisphenol A polyoxyethylene ether.
These bisphenol A/ethylene oxide and bisphenol A/propylene oxide polyethers can be used in polyurethane elastomers, polyurethane powder coatings, UV-curing polyurethane acrylate coatings, adhesives, polyurethane and PIR foam plastics, etc. The materials exhibit good toughness, hardness, flame retardancy, heat resistance, and water resistance. These aromatic polyols are also used in epoxy resins, acrylates, and polyester resins.
Mannich Polyether Polyols
Aromatic polyethers with a functionality greater than 3 can be easily prepared from aromatic diamines such as toluenediamine. Some rigid foam polyethers are such products. For example, the rigid foam polyether TDA-401 from the Polyether Department of Sinopec Tianjin Petrochemical Company is a polyoxypropylene polyol initiated with triethanolamine and toluenediamine, with a hydroxyl value of 385-415 mg KOH/g, a viscosity of 9-17 Pa·s, and a functionality of approximately 3.7.
The Mannich reaction is an important method for preparing rigid foam polyether polyols. The Mannich reaction involves a three-component condensation reaction between a compound containing an active hydrogen (such as a phenol), an aldehyde (usually formaldehyde), and a basic amine (primary or secondary amine). The active hydrogen is replaced by an aminomethyl group, and the resulting product is called a Mannich base. This reaction is also known as aminomethylation. In polyether production, aromatic amine polyols (Mannich bases) are prepared using phenol (or nonylphenol, etc.), formaldehyde, and a diolamine as raw materials via the Mannich reaction. Subsequent ring-opening polymerization of alkylene oxides yields polyether polyols containing benzene rings, also known as Mannich polyols. By controlling the functionality of the initiator and the amount of alkylene oxide used, the hydroxyl value and functionality of the polyether polyol can be adjusted, with the functionality generally ranging from 3 to 7. The polyether contains aromatic rings with aldehyde structures from the phenol-formaldehyde reaction and tertiary amino groups. The introduction of benzene rings provides polyurethane products with excellent heat resistance, flame retardancy, and mechanical properties, while the tertiary amino groups provide a certain degree of self-catalysis, allowing for the use of less or no catalyst in foaming formulations.
Some manufacturers produce rigid foam polyethers using this method. For example, Fangda Jinhua Chemical Technology Co., Ltd.’s rigid foam polyether JH-4548 is synthesized using a composite initiator of glycerol, Mannich base, sucrose, and sorbitol, with a hydroxyl value of 440–480 mg KOH/g, a pH of 9–11, and a viscosity of 1700–2400 mPa·s. The Mannich base (initiator) is prepared from phenol, bisphenol A, diethanolamine, and formaldehyde. Additionally, Sinopec Tianjin Petrochemical Company’s polyether division produces the spray-applied rigid foam polyether polyol SY-6560 using a Mannich initiator followed by polymerization with PO. It has a functionality of approximately 6, a hydroxyl value of 540–600 mg KOH/g, and a viscosity of 2000–2400 mPa·s.
PREVIOUS:Polytrimethylene Ether Glycol
CATEGORIES
LATEST NEWS
- Diethanolamine
- Triethanolamine
- Methyl Glucoside
- Mannitol
- Sorbitol
- Xylitol
- Pentaerythritol
- Trihydroxyethyl isocyanurate
- Trimethylolethane
- Glycerin
CONTACT US
Tel:+8618688691634
Email:sam@1688pu.com
Add:No. 91 Central East Rd, Luxi village, Gaobu town, Dongguan city, Guangdong Province, China

