Ethylene Glycol

Alias: Glycol, Monoethylene Glycol.
Abbreviation: EG, MEG.
Molecular formula: C2H6O2
Molecular weight: 62.1
CAS number: 107-21-1
EINECS number: 203-473-3
Structural formula: HOCH2CH2OH.
Physicochemical Properties
A colorless, transparent, viscous liquid with a sweet taste, hygroscopic, and flammable. Explosion limits: 3.2%–15.3% (volume fraction). Ethylene glycol is miscible with water, lower aliphatic alcohols, glycerol, acetic acid, ketones, aldehydes, and pyridine; slightly soluble in diethyl ether; and almost insoluble in benzene and its homologs, chlorinated hydrocarbons, petroleum ether, and oils. It significantly lowers the freezing point of water. Typical physicochemical properties of ethylene glycol are shown in Table 5-13.
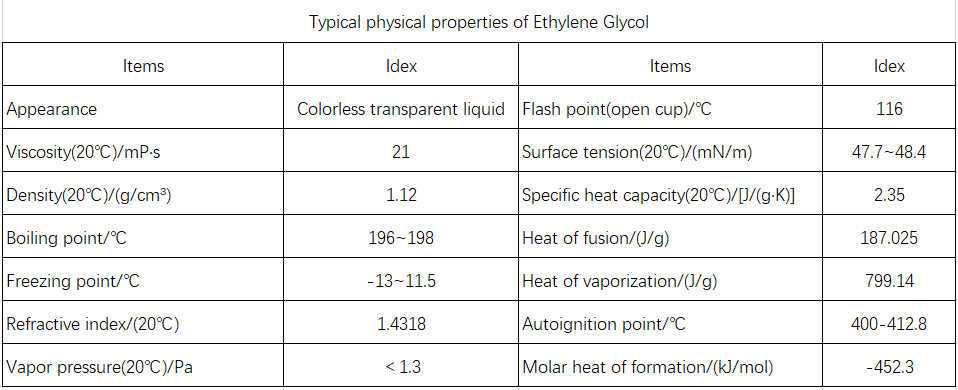
Characteristics and Uses
Mainly used as a raw material for the polymerization of terephthalic acid (PTA) to produce PET polyester resin and fibers (polyester); also used in polyester polyols, unsaturated polyester resins, etc. It can also be used as a solvent and in the preparation of engine antifreeze, and in the manufacture of cosmetics and explosives (dinitroglycol).
In the polyurethane field, ethylene glycol is mainly used in the synthesis of polyester polyols, and can also be used as an initiator to synthesize polyether diols, polyurethane chain extenders, etc.
Toxicity
Ethylene glycol has very low toxicity. The reported lowest lethal dose (human) LDL0 = 786 mg/kg, and the acute oral toxicity value for rats LD50 = 4.7–8.5 g/kg. Due to its high boiling point and low vapor pressure, inhalation poisoning is generally not a concern.
PREVIOUS:ε-Caprolactone
NEXT:1,4-Butanediol
CATEGORIES
LATEST NEWS
- Diethanolamine
- Triethanolamine
- Methyl Glucoside
- Mannitol
- Sorbitol
- Xylitol
- Pentaerythritol
- Trihydroxyethyl isocyanurate
- Trimethylolethane
- Glycerin
CONTACT US
Tel:+8618688691634
Email:sam@1688pu.com
Add:No. 91 Central East Rd, Luxi village, Gaobu town, Dongguan city, Guangdong Province, China

