Ethylene Oxide

Synonyms: Ethylene oxide
Abbreviation: EO
Molecular formula: C2H4O
Molecular weight: 44.06
CAS number: 75-21-8
EINECS number: 200-849-9
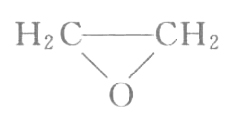
Structural formula:
Physicochemical Properties
At room temperature, it is a colorless gas, which can be liquefied into a transparent liquid under pressure or cooling. Boiling point: 10.4℃, Flash point: -17℃, Liquid density (20℃): 0.87 g/mL, Freezing point: -113℃, Vapor pressure (20℃): 151.6 kPa. Critical pressure: 7.19 MPa, Critical volume: 0.00319 m³/kg. Vapor density is 1.5 times that of air. Ethylene oxide forms explosive mixtures with air, with an explosion limit of 3% to 100% (volume fraction). It is soluble in water and readily reacts with compounds containing active hydrogen. Ethylene oxide vapor is highly toxic, and the acute toxicity data for rats shows an LD50 of approximately 100 mg/kg.
Characteristics and Uses
The hydration of ethylene oxide yields ethylene glycol, which is a raw material for resins such as polyesters and polyurethanes.
In the polyurethane field, ethylene oxide is used to manufacture hydrophilic or highly reactive polyether polyols, and also for the manufacture of silicone-polyether copolymer foam stabilizers, etc.
A large amount of ethylene oxide is also used in the manufacture of surfactants, etc. Gaseous ethylene oxide can be used as a disinfectant.
Ethylene oxide is highly reactive, and the reaction process is strongly exothermic. Therefore, great care must be taken when handling ethylene oxide, and attention should be paid to its toxicity, flammability, instantaneous polymerization, and high-temperature decomposition characteristics. Safety measures should be taken.
PREVIOUS:Propylene Oxide
NEXT:Tetrahydrofuran
CATEGORIES
LATEST NEWS
- Diethanolamine
- Triethanolamine
- Methyl Glucoside
- Mannitol
- Sorbitol
- Xylitol
- Pentaerythritol
- Trihydroxyethyl isocyanurate
- Trimethylolethane
- Glycerin
CONTACT US
Tel:+8618688691634
Email:sam@1688pu.com
Add:No. 91 Central East Rd, Luxi village, Gaobu town, Dongguan city, Guangdong Province, China

