Glycerin

Chemical Name: 1,2,3-Propanetriol; Glycerol.
Abbreviation: GLY.
Synonym: 1,2,3-Trihydroxypropane.
Molecular Formula: C₃H₈O₃
Molecular Weight: 92.1
CAS No.: 56-81-5
EINECS No.: 200-289-5
Structural Formula:
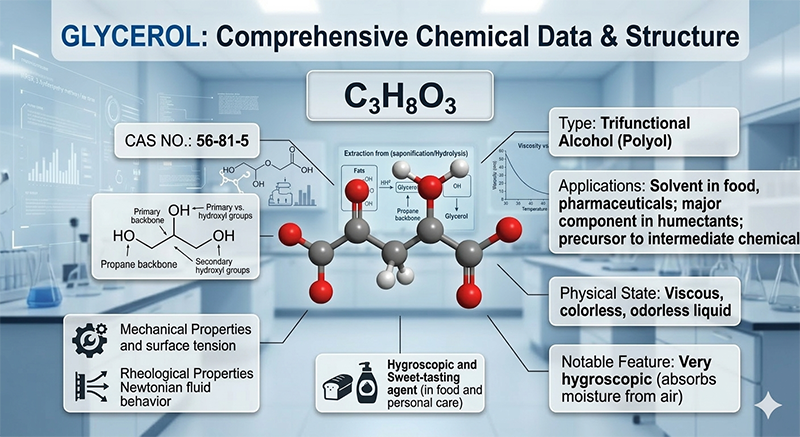
Physicochemical Properties:
A colorless, transparent, odorless, and viscous liquid with a sweet taste. It possesses strong hygroscopic properties and exhibits low toxicity. It is miscible with water and ethanol, and is capable of lowering the freezing point of water. It is slightly soluble in ethyl acetate and diethyl ether, but insoluble in benzene, chloroform, carbon tetrachloride, petroleum ether, and oils.
Pure glycerin has a relative density of 1.261, a surface tension (at 20°C) of 64.0 mN/m, a refractive index of 1.473, a flash point of 160°C, a melting point of 18°C, a boiling point of 290°C, and an autoignition temperature of 370°C. Its vapor pressure is very low at room temperature; specifically, the vapor pressure is <0.1 Pa at 20°C and 0.33 Pa at 50°C.
Industrial-grade glycerin with a purity of ≥95% has a relative density (at 20°C) of ≥1.248, a viscosity (at 20°C) of ≥50 mPa·s, and a refractive index (at 20°C) ranging from 1.440 to 1.470. It demonstrates heat resistance: when subjected to (120 ± 5)°C for 3 minutes, it neither decomposes nor discolors. It also exhibits cold resistance: when subjected to (-10 ± 2)°C for 24 hours, it neither crystallizes nor separates into layers. Its pH value ranges from 6 to 8.
Upon heating with loss of water, it forms diglycerol, polyglycerols, and similar compounds; upon oxidation, it yields glyceraldehyde, glyceric acid, and related substances; and upon reduction, it produces propylene glycol. It undergoes decomposition at temperatures below its boiling point, with a decomposition temperature of 171°C. When heated to temperatures exceeding 280°C, glycerin decomposes to generate acrolein—a substance characterized by its strong toxicity and corrosiveness.
Properties and Applications:
In the pharmaceutical sector, it is used to produce various preparations, solvents, humectants, antifreeze agents, and sweeteners, as well as to formulate topical ointments, suppositories, and similar products. Nitroglycerin, synthesized from glycerin, serves as a raw material for explosives. It is a crucial raw material for synthetic resins, utilized in the manufacture of alkyd resins, polyester resins, glycidyl ethers, polyether polyols, and epoxy resins. In textile and dyeing processes, it is employed to produce wetting agents, humectants, anti-wrinkle and anti-shrinkage treatment agents for fabrics, as well as dispersing and penetrating agents. In the instrumentation industry, it functions as a sweetener, a humectant for tobacco products, and a solvent. Furthermore, it finds widespread application in industries such as papermaking, cosmetics, leather tanning, photography, printing, metal processing, and rubber manufacturing.
In the field of polyurethanes, its most significant application is as an initiator for polyether polyols—specifically polyether triols—though it is also utilized in the production of polyurethane foams, coatings, and adhesives.
Toxicity:
Glycerin is considered virtually non-toxic; acute oral toxicity data for rats indicates an LD50 of 12.6 g/kg. Repeated skin contact may lead to skin dehydration, while inhalation of high concentrations of its mist or fumes may cause irritation; the typical hazardous threshold limit value is 10 mg/m³.
PREVIOUS:Trimethylolpropane
NEXT:Trimethylolethane
CATEGORIES
LATEST NEWS
- Diethanolamine
- Triethanolamine
- Methyl Glucoside
- Mannitol
- Sorbitol
- Xylitol
- Pentaerythritol
- Trihydroxyethyl isocyanurate
- Trimethylolethane
- Glycerin
CONTACT US
Tel:+8618688691634
Email:sam@1688pu.com
Add:No. 91 Central East Rd, Luxi village, Gaobu town, Dongguan city, Guangdong Province, China

