Tetramethyl-m-xylylene diisocyanate

Abbreviation:TMXDI, m-TMXDI
Synonyms: Tetramethyl-m-xylylene diisocyanate, 1,3-bis(1-isocyanato-1-methylethyl)benzene.
Molecular formula: C14H16N2O2
Molecular weight: 244.29.
The CAS number for m-TMXDI is 2778-42-9, 58067-42-8, and the EINECS number is 220-474-4. The CAS number for p-TMXDI (tetramethyl-p-xylylene diisocyanate) is 2778-41-8, and the EINECS number is 220-473-9.
Structural Formula
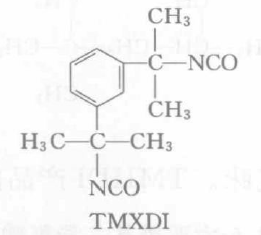
Tetramethylxylylene diisocyanate has two isomers, meta and para. m-TMXDI is already produced industrially, while p-TMXDI is not yet commercially available.
Physical and Chemical Properties
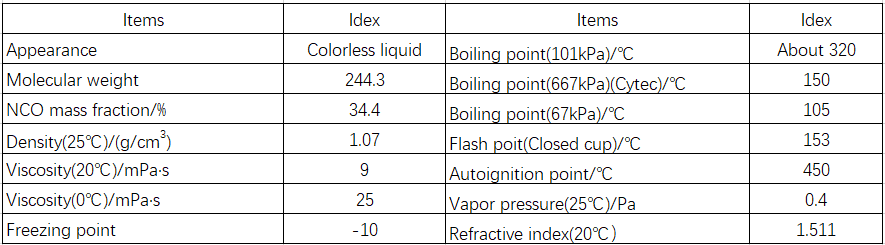
m-TMXDI is a colorless liquid at room temperature, with a freezing point of -10°C. It is soluble in most polar organic solvents and slightly soluble in aliphatic hydrocarbons. It is insoluble in water and reacts with water, alcohols, and amines. Typical physical properties of m-TMXDI products are shown in Table 1-24.
In addition, p-TMXDI is a white crystalline solid at room temperature, with a melting point of 72°C.
Characteristics and Uses
The molecular structure of tetramethylxylylene diisocyanate is such that the hydrogen atoms on the two methylene groups of XDI are replaced by methyl groups, and the NCO groups are on the methylene groups connected to the benzene ring, not conjugated with the aromatic ring. Therefore, it possesses characteristics of both aliphatic and aromatic compounds, resulting in elastomers that are soft and have high strength, adhesion, appearance, flexibility, and durability.
The methyl substitution of hydrogen atoms improves UV aging resistance and hydrolysis stability, weakens hydrogen bonding, and increases the elongation of polyurethanes. Furthermore, the two NCO groups of TMXDI are tertiary NCO groups, and due to steric hindrance, the reactivity of the NCO groups is reduced. Its low reactivity allows for emulsification at higher temperatures and results in lower prepolymer viscosity, making it particularly suitable for the preparation of waterborne polyurethanes without the need for organic solvents. It can be used in the production of solvent-free waterborne polyurethane adhesives and coatings, including waterborne polyurethane automotive primers, waterborne plastic coatings and wood coatings, waterborne inks, low-temperature activated waterborne polyurethane adhesives, lamination adhesives, and waterborne polyurethane leather finishes.
TMXDI can also be used to prepare blocked isocyanates, which can then be used to formulate one-component thermosetting coatings. The deblocking temperature of tertiary isocyanates is 10-15°C lower than that of primary and secondary isocyanates.
Toxicity and Protection:
TMXDI is a low-toxicity diisocyanate. The acute oral toxicity LD50 in rats is 5000 mg/kg, the acute dermal toxicity LD50 in rabbits is ≥2000 mg/kg, the inhalation toxicity LC50 in rats is 2.7 mL/(m³.4h) or 0.027 mg/(L.4h), and the acute inhalation toxicity LC50 in guinea pigs for 1 hour is 0.240 × 10⁻⁶.
TMXDI vapor is irritating and toxic by inhalation. The liquid may be irritating to the eyes and can cause skin allergic reactions. The workplace should be well-ventilated, and protective gloves and work clothing should be worn.
CATEGORIES
LATEST NEWS
- Diethanolamine
- Triethanolamine
- Methyl Glucoside
- Mannitol
- Sorbitol
- Xylitol
- Pentaerythritol
- Trihydroxyethyl isocyanurate
- Trimethylolethane
- Glycerin
CONTACT US
Tel:+8618688691634
Email:sam@1688pu.com
Add:No. 91 Central East Rd, Luxi village, Gaobu town, Dongguan city, Guangdong Province, China

